MENU
FR | EUR
-
-
-
-
- WHX Labs Dubai 2026
- analytica 2026
- Forum Labo 2026
- SLAS Europe 2026
- Forum Labo 2026 Bioprocess
- Bioprocessing Summit Europe 2026
- BioProcess International 2026
- Swiss Biotech Day 2025
- ISCT 2026
- Pichia 2026
- Future Labs Live 2026
- BioProScale Sympposium 2026
- ASGCT 2026
- Eppendorf Automation Forum 2026
- ESACT 2026
-
-
-
-
- WHX Labs Dubai 2026
- analytica 2026
- Forum Labo 2026
- SLAS Europe 2026
- Forum Labo 2026 Bioprocess
- Bioprocessing Summit Europe 2026
- BioProcess International 2026
- Swiss Biotech Day 2025
- ISCT 2026
- Pichia 2026
- Future Labs Live 2026
- BioProScale Sympposium 2026
- ASGCT 2026
- Eppendorf Automation Forum 2026
- ESACT 2026
FR | EUR
-
- Toutes les centrifugeuses
- Centrifugeuses de paillasse
- Centrifugeuses au sol
- Centrifugeuses réfrigérées
- Microcentrifugeuses
- Centrifugeuses polyvalentes
- Centrifugeuses haute vitesse
- Ultracentrifugeuses
- Concentrateur
- Produits IVD
- Consommables pour centrifugeuses haute vitesse et ultracentrifugeuses
- Tubes à centrifuger
- Plaques de centrifugeuse
- Logiciel de gestion d’appareils
- Gestion des échantillons et des informations
-
- Pipettes, distributeurs et dispositifs de manipulation de liquides automatisée
- Pipettes mécaniques
- Pipettes électroniques
- Pipettes multicanaux
- Pipettes et distributeurs à déplacement positif
- Pointes de pipette
- Distributeurs sur flacon
- Auxiliaires de pipetage
- Accessoires pour pipettes et distributeurs
- Pipetage automatisé
- Consommables d’automatisation
- Accessoires d’automatisation
- Services pour pipettes et distributeurs
Sorry, we couldn't find anything on our website containing your search term.
Sorry, we couldn't find anything on our website containing your search term.

Vaccine Research Discovered
Well thought-out tools for pharmaceutical and biotech laboratories
Read more
Read less
TRADITONAL VACCINES
Inactivated vaccines
Inactivated vaccines (e.g., for hepatitis A, rabies, polio) still carry the full repertoire of immunogenic components of the original pathogen. Proper inactivation is mandatory to avoid viral reactivation and replication in the host.
➕ Pathogen inactivation by radiation, heat, or chemical reagents ensures a superior safety profile and advantages in terms of transport and storage.
➖ Require processing of large quantities of the pathogen. The inactivation process can affect the antigen immunogenicity. As the antibody titers reduces over time, several booster doses are needed.
Inactivated vaccines (e.g., for hepatitis A, rabies, polio) still carry the full repertoire of immunogenic components of the original pathogen. Proper inactivation is mandatory to avoid viral reactivation and replication in the host.
➕ Pathogen inactivation by radiation, heat, or chemical reagents ensures a superior safety profile and advantages in terms of transport and storage.
➖ Require processing of large quantities of the pathogen. The inactivation process can affect the antigen immunogenicity. As the antibody titers reduces over time, several booster doses are needed.
Lire la suite
Live-attenuated vaccines
Repeated subculturing in a foreign host belongs to the most common methods to obtain attenuation of live-attenuated vaccines (e.g., for variola virus, measles virus, rotavirus). With each passage, the virus becomes less virulent. The native viral antigen conformation is still preserved, mimicking the natural infection without causing disease.
➕ Usually produce long-lasting robust cellular and humoral immune responses with only one administration.
➖ Safety issues in immunosuppressed people may occur, weakened strains are difficult to obtain, development time is long, and refrigeration obligatory.
Repeated subculturing in a foreign host belongs to the most common methods to obtain attenuation of live-attenuated vaccines (e.g., for variola virus, measles virus, rotavirus). With each passage, the virus becomes less virulent. The native viral antigen conformation is still preserved, mimicking the natural infection without causing disease.
➕ Usually produce long-lasting robust cellular and humoral immune responses with only one administration.
➖ Safety issues in immunosuppressed people may occur, weakened strains are difficult to obtain, development time is long, and refrigeration obligatory.
Lire la suite
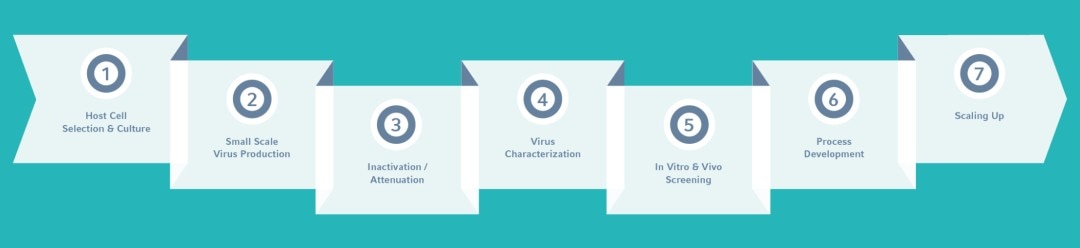
IMPROVE THE DEVELOPMENT OF VIRAL INACTICATED / ATTENUATED VACCINES
Lire la suite
*All products are for Research Use Only (RUO).
Lire la suite
DOWNLOADS

Application Note

Application Note

Application Note

White Paper

White Paper
Lire la suite